HIV is one of the deadliest viruses encountered by humans in recent history and will kill 1.5m people this year alone, compare this to Ebola, for example, which has killed less than 5,000 so far, and you get an idea of how big a problem HIV still is. Such an obvious foe to humanity would seem to be an unlikely choice to save lives, then, but increasingly it is being used to tackle another killer disease – cancer.
A new era in the understanding of our DNA was kick-started by the completion of the human genome project in 2001, giving us more insight than ever before into our DNA and how it can cause disease when something goes wrong with it. Cancer is universally caused by abnormalities in DNA, some of which are the result of preventable triggers such as smoking, but many of which may be the consequence of a small risk of mistakes every time our infinitely complicated DNA is copied as part of normal cell growth.
Immortal cells
Researchers frequently use samples from people with cancer to find out more about the disease but these often quickly perish in the lab from being outside the body. To overcome this, researchers have an arsenal of different types of “immortal cells” called cell lines, which keep dividing and growing over time and have special modifications in their DNA to allow them to live in artificial conditions that mimic the human body in the laboratory.
There are cell lines from cancers of the blood, breast and lung among many others, but these often have countless numbers of DNA abnormalities and it can be difficult to pinpoint which one, or combination of abnormalities, is responsible for making the cell cancerous. Scientists often want to look at specific genetic changes, which they think contribute to a healthy cell becoming a cancer cell – and this is where HIV can be utilised.
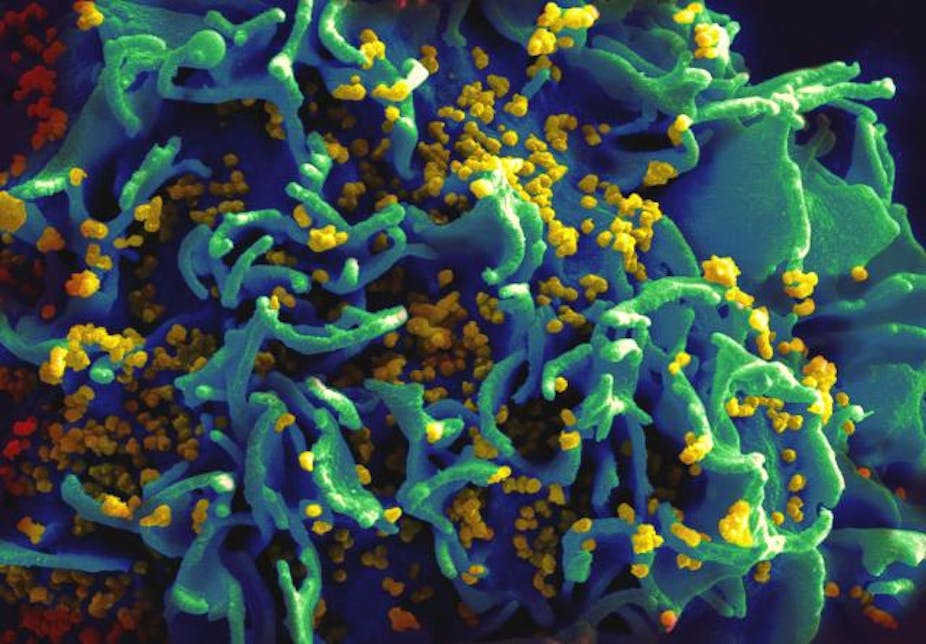
Like many viruses, HIV functions by inserting its DNA, the genetic code which is the blueprint for its structure and actions, into the DNA of human cells. If successful, this viral DNA is then indistinguishable from human DNA to the cell, meaning that when the cell divides, it dutifully copies not just its own DNA but the viral DNA too, creating more viruses.
Hijacking HIV
As exploitative as this process appears, scientists are now able to hijack this unique viral ability to transfer genetic information of their choosing into cells. In just a few days in a well-equipped laboratory, scientists can now cut, copy and paste cancer-contributing DNA into HIV, harvest the virus and infect immortal cell lines, creating an unlimited reservoir of cells which have only a single DNA change compared to non HIV-infected cells. Scientists can then see exactly what the specific piece of abnormal DNA does to the cells, for example how it changes the resilience of the cells to a new chemotherapy drug.
This new-found role of HIV does not end in the laboratory. Last year, online articles and videos circulated widely on social media, claiming that doctors had “cured” an eight-year-old girl called Emily Whitehead of leukaemia (blood cancer) by “injecting her with HIV”. This misleading and sensationalist headline obscured what was actually an incredibly promising new avenue in treating cancer.

T-cells, types of white blood cell, were taken from Emily and infected with modified HIV. The virus obediently transferred its DNA into Emily’s T-cells, but scientists had also deleted the DNA which would normally allow it to replicate, removing the possibility that Emily herself could get HIV from the treatment. This modified HIV carried DNA that allowed the T-cells to track down the leukaemic white blood cells inside Emily’s body and kill them. Emily remains disease free at the time of writing, over two-and-a half-years after treatment.
Destabilising effects
A major problem in treating cancer is that there are more than 200 different types – and even within a single one of these, leukaemia for example, there are many hundreds of different DNA abnormalities which cause the disease. Effectively, this means that in an ideal world, most people with cancer need “personalised treatments”, which is currently not possible most of the time.
What is unique about the HIV-based treatment is that if the differences between cancer cells and healthy cells can be identified in each patient, HIV can be used to mobilise the immune system against a wide range of these abnormal cells and designed to suit the needs of individual patients.
Notwithstanding early successes with Emily and a handful of other individuals, the treatment is still in the early stages of development, with many hurdles to overcome before it can truly be considered a major breakthrough. Too many T-cells can cause dangerous side effects by destabilising the normal functions of the immune system. Despite eventually recovering, Emily was unfortunate enough to experience these.
Leukaemia is also a comparatively easy cancer to target, as many of the cancerous cells are in the blood and easily reached by therapies injected directly into the bloodstream, whereas in cancerous tumours, many cells are more difficult to reach. Although promising, and definitely innovative, it remains to be proven whether HIV-based therapies can overcome this longstanding problem.
Years of research into HIV may not have been able to eradicate it yet, but it has resulted in a most unlikely and welcome ally in the fight against cancer.