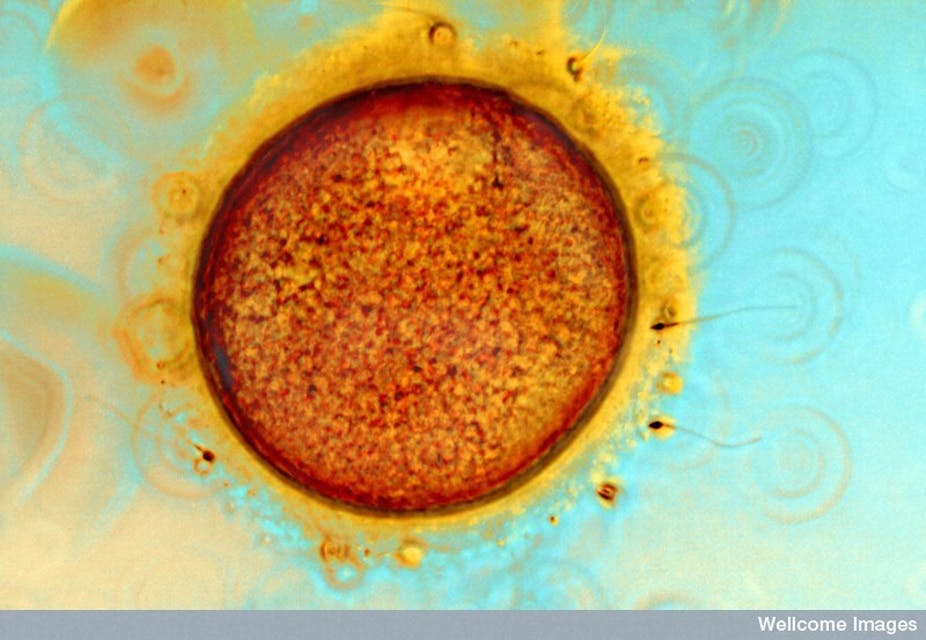
A new method of predicting the chances an embryo will survive has shown promise in tests on mice and could one day be used on humans to boost IVF success rates, UK scientists have said.
Many couples undergoing in vitro fertilisation find that several rounds of the expensive and invasive procedure are required for successful conception to occur because it can be difficult to know which embryos will go on to form a viable pregnancy.
Some women opt to have several embryos implanted at once, which can lead to risky multiple pregnancies.
In a paper published in the journal Nature Communications, UK researchers report observing a link between the way an egg pulsates shortly after fertilisation and the likelihood the resulting embryo will survive.
“This method offers, thus far, the earliest and fastest, non-invasive way to predict the viability of eggs fertilised in vitro and therefore can potentially improve greatly the prospects for IVF treatment,” the paper said.
Lead author Professor Magdalena Zernicka-Goetz of the Department of Physiology, Development and Neuroscience at Cambridge University said the sooner a viable embryo was identified, the better.
“It is important to be able to quantitate some indication of the health of embryos at the earliest possible stage in order to minimise the time they have to spend outside the body of the mother,” she said.
The researchers took time-lapse photographs of pulsating mouse eggs two hours after fertilisation and found the speed of the movement was linked to the likelihood the embryo would develop. Eggs with faster movements and those with a longer interval between movements developed most successfully. They then built a mathematical model to predict the chances the embryo would survive gestation.
Dr Allan Pacey from the University of Sheffield’s Academic Unit of Reproductive and Developmental Medicine said the technique would have to be simplified before it could be used in commercial IVF labs.
“This is the biggest hurdle I anticipate that might prevent more people experimenting with this approach,” said Dr Pacey, who was not involved in the research.
“But I hope they do, as we really need to develop something more technical than the ‘watch and wait’ approach we have currently.”
Dr Robert Norman, professor of reproductive and periconceptual medicine at the University of Adelaide and director of the Robinson Institute, said the technique was interesting scientifically but a long way from becoming a reality for couples undergoing IVF.
“It has to be confirmed in humans and upscaled to be able to monitor a lot of eggs. And the sort of equipment you would need to put this into routine use is enormous,” he said.
“But this is in the front running for being right of the top of what we are interested in right now.”