Bovine tuberculosis is a major problem in the UK: in 2013 around 8m cattle were tested and 32,000 slaughtered at a cost of an estimated £100m, including compensation – a huge economic burden that makes controlling the disease essential.
However, far from controlling bovine TB (bTB), over the past 20 years or so there has been a 10% increase in cases each year. This rise, and the role of TB-infected badgers, is fiercely debated. Badgers were first associated with bTB in the 1970s, but despite the lengthy and expensive Randomised Badger Culling Trial in the late 1990s there is still limited scientific consensus.
One of the main difficulties in trying to untangle the routes through which bTB is transmitted is the fact that it spreads unseen. In general, infected animals are only identified through a skin test, which in itself we estimate to be only 72% sensitive – so 28% of infectious animals are undetected by a single test. With more than a quarter of infected animals going undetected, controlling the disease’s spread is difficult, and identifying how it spreads harder still.
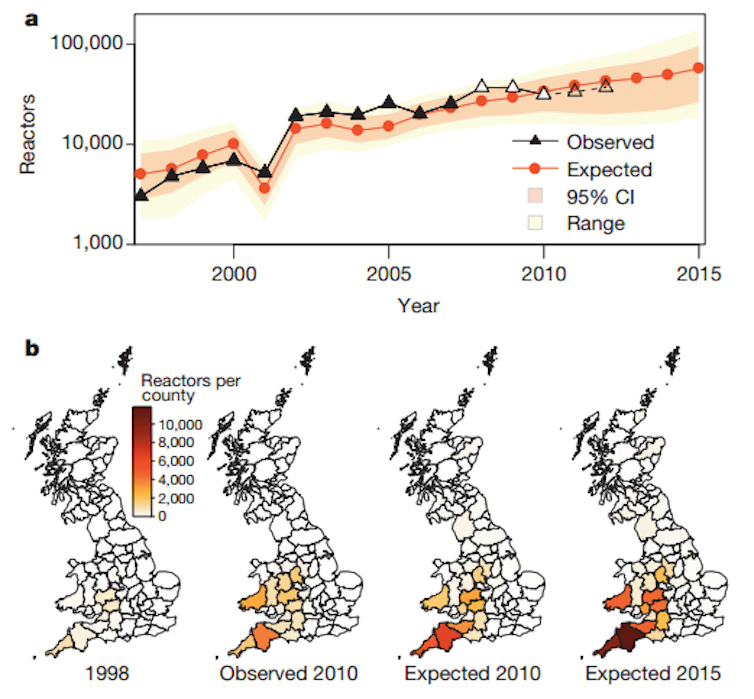
Published in Nature, we have developed a mechanistic model of the spread of bTB within and between around 134,000 cattle farms in Great Britain, between 1996 and 2011. This model accounts for multiple routes of transmission: direct infections between cattle, indirectly through the environment, and through the movement of infected animals between farms. Because the model is mechanistic it allows us to test the effects of different methods of control on how the disease spreads.
Building a model
Due to the lack of detailed data, our model does not explicitly include badgers (cattle are ear-tagged, their movements recorded, and are frequently tested for bTB; the same can’t be done for wildlife). Instead we model the combined environmental reservoir of infection – this includes infection from pasture and wildlife species, although it is impossible to separate these two elements.
We don’t feel that this detracts from our findings, as the model is able to accurately capture the trends of infection over time and the geographic spread. We haven’t found any evidence that our model underestimates the rate of infection in regions where badgers are common, which would occur if the model underestimated their importance.
Our modelling approach was able to shed light on the relative strengths of different transmission routes. Unlike the real world where infections can only be seen by testing (and then with limited accuracy) within our simulation model we can track infection perfectly. This allows us to tease apart the causes of infection.
We find that newly infected farms are most frequently due to movement of infected animals, rather than spread through the environmental reservoir, but most importantly the majority of infection originates from the minority of farms. More than 90% of infected farms clear the infection before they cause any secondary cases, while about 2% of farms infect 10 or more other farms.
Our model shows just how complex and multifaceted bTB transmission is, with cattle-to-cattle transmission, failure to detect infectious cases, the movement of infected animals to other farms, and the effects of TB in the environment all playing a role. It is this complexity that continues to deny those seeking to eradicate the disease any easy solutions.
The way forward
We tested a range of control measures in our model simulation to see what effects they might have. We assumed that these measures began in 2005 and were continued for six years, after which we evaluated their impact. Of ten we simulated, only three measures were found to stop the annual increase in cases.
By far the most dramatic is whole-herd culling of all cattle on infected farms. This would rapidly bring the epidemic under control, as it removes the danger of infected but as yet undetected animals. But it would entail a 20-fold increase of cattle slaughter in the first year, and the political, social, and economic consequences that would bring. However, with the cost of bTB at £100m a year and rising, some form of highly targeting culling could be economically viable.
Vaccinating cattle, which slows the disease’s progression in animals, is also predicted sufficient to prevent the increase in cases, although only just. Such a vaccine would not have the effectiveness we associate with mass vaccination programmes in humans. In any case, the vaccine is currently unlicensed and runs into problems with EU legislation.
More widespread and regular testing and slaughter of infected animals in the initial years would identify more cases (we only find bTB when we look for it, after all) and lead to more slaughter, but over time the benefits of a more stringent regime are felt and cases fall in later years. A similar effect can be achieved if we developed a better test for bTB.
What the model also predicted was the relatively limited effect of focusing on environmental factors would be. Even with a 50% fall in the between-farm transmission of infection due to the environment (simulating a substantial badger cull, for example), the rise in cattle infections over six years continues, although at a slower rate.
All three of these measures (culling, increased testing and cattle vaccination) have associated economic costs and benefits, which the government, the livestock industry and the general public must assess to decide the correct course of action. What our simulation provides, based on the latest data, is a way to predict and put numbers to a range of potential policy options. Clearly there is a need to reverse this costly and damaging bTB epidemic, and we hope our findings will help inform decisions in the future.

