As a graduate student in the 1970s, microbiologist Richard Blakemore probably wasn’t expecting to discover a new bacterial species with a never-before-seen ability. While studying bacteria that live in muddy swamps, he observed that some tended to swim reliably toward the same geographical direction. Even when he rotated the microscope, they persisted in wiggling toward one direction. After confirming that their swimming behaviors were unaffected by light, Blakemore suspected they might be responding to the weak magnetic fields naturally present on Earth.
After further tests and observations, Blakemore confirmed the bacteria were reacting to magnetism. He published a landmark paper in the journal Science in 1975 introducing magnetotactic bacteria to the wider world. Later, researchers realized that another scientist, Salvatore Bellini, had previously discovered magnetotactic bacteria, but his work received scant attention because it hadn’t been archived.
In the decades since, scientists have continued to study how these tiny magnetic creatures behave. Of course it’s just cool to learn more about these unique single-celled organisms. But researchers like me are also figuring out ways to harness their magnetic properties in medical and other engineering applications.
What makes them living magnets?
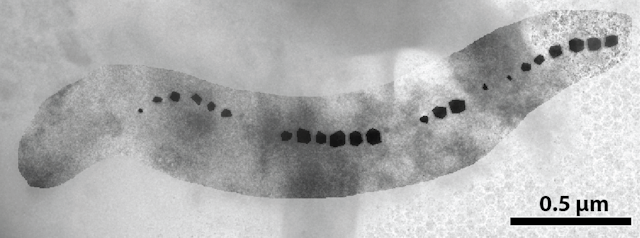
You’ve probably stuck a magnet to the metal door of a refrigerator before. This unique group of prokaryotes basically contain super tiny versions of those fridge magnets. They pack either iron-oxide or iron-sulfide molecules into highly dense structures known as magnetic nanoparticles.
Each nanoparticle is about 100,000 times smaller than a grain of rice. Magnetotactic bacteria produce them in different shapes: bullet, rectangular and spherical. Researchers aren’t sure of a reason for this variation, but a possible explanation is that differently shaped particles can interact differently with magnetic fields.
By clustering and aligning in chains, these magnetic nanoparticles enable magnetotactic bacteria to respond even to the weak magnetic fields of the Earth – a strength of about 0.5 Gauss, as opposed to the 100 Gauss of a refrigerator magnet.
Where did magnetotactic bacteria come from?
There are two main proposals for how magnetotactic bacteria emerged on Earth.
The first hypothesis suggests that this group of bacteria evolved a couple billion years ago, in a time of increasingly abundant oxygen. As the oxygen reacted with iron, the amount of iron dissolved in the oceans decreased.

Living things need iron for metabolic activities such as respiration, so bacteria started storing it to prevent coming up short in times of scarcity. But high concentrations of freely diffusing iron are toxic for cells.
The idea is that evolution favored bacteria that wound up crystallizing iron into nanoparticles and wrapped a lipid membrane around them to form magnetosomes.
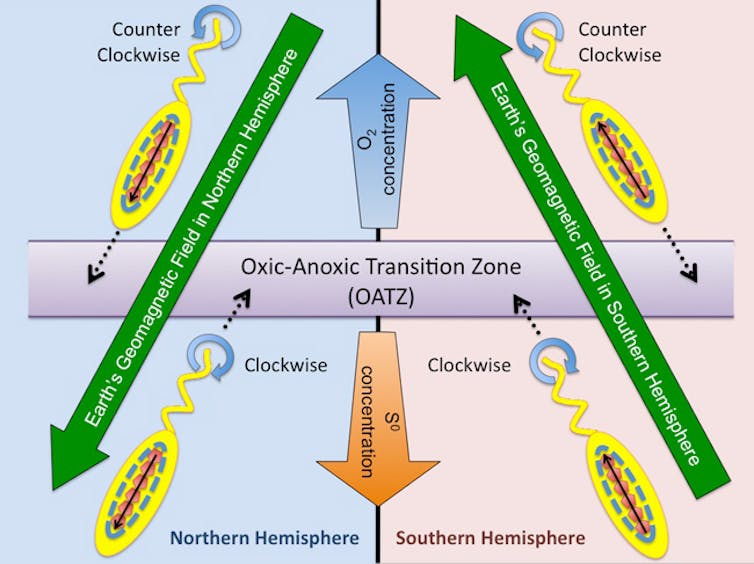
An alternative explanation is more widely accepted by biologists. It’s based on the observation that magnetotactic bacteria grow best in environments like the swamps where they were first discovered – places with very limited oxygen, at concentrations as low as 1 to 2 percent.
As a magnetotactic bacterium moves through a swampy bog, it’s likely to encounter sand or soil particles that could obstruct its path. A bacterium can actively use its flagellum – a whip-like appendage that propels it while swimming - to move past these sediments to reach its preferred growth environment.
But in some cases, the flagellum might not be powerful enough. Magnetic particles can provide some additional force for these bacteria, allowing them to make use of Earth’s magnetic field for navigation and a little extra thrust forward. Magnetosomes allow for more effective navigation.
Isolating and using magnetic genes in the lab
For many years, scientists have been trying to determine whether animals including bees, sea turtles, sharks and pigeons are magneto-sensitive. Could this possible sense – called magnetoreception – help them with amazing feats of navigation? So far studies have been mostly inconclusive.
Studying simpler organisms like the magnetotactic bacteria might be one way to better understand how genes regulate biomagnetism.
By creating mutations in the lab, microbiologists [have identified genes] that enable magnetotactic bacteria to produce magnetic nanoparticles. They’ve also found genes that control the nanoparticles’ size, shape and alignment in these bacteria.
One possible application is to use these magnetic genes as a tool to manipulate cells in a non-invasive way. They could allow a researcher to wirelessly control a cell.
Magnetogenetics could build on the technique of optogenetics, a method that uses light to precisely manipulate cell activities. For instance, a researcher can trigger a genetically engineered neuron to fire by exposing it to light. Light cannot penetrate very far through tissue, though, so it can’t get into deep brain regions or the gut, for instance.
Magnetic fields, on the other hand, easily penetrate bodily tissues. By engineering magnetic cells and manipulating them, scientists hope to learn more about the functions of specific cell types. Ultimately this knowledge could help treat diseases.
Scientists haven’t yet had any success in creating magnetic cells, except in one strain of photosynthetic bacterium. Reports of creating magnetic mammalian cells are controversial. So far they only contain super-tiny magnetic nanoparticles that are randomly distributed in the cells.
My colleagues and I worked on a way to help figure out which magnetism-related mutations might be useful. First, we used chemicals to randomly generate mutant bacteria with different numbers of magnetic nanoparticles. Then, using a magnetic device we developed that has unprecedented sensitivity, we were able to sort and separate mutants with no nanoparticles and those with up to three times more than the normal number.
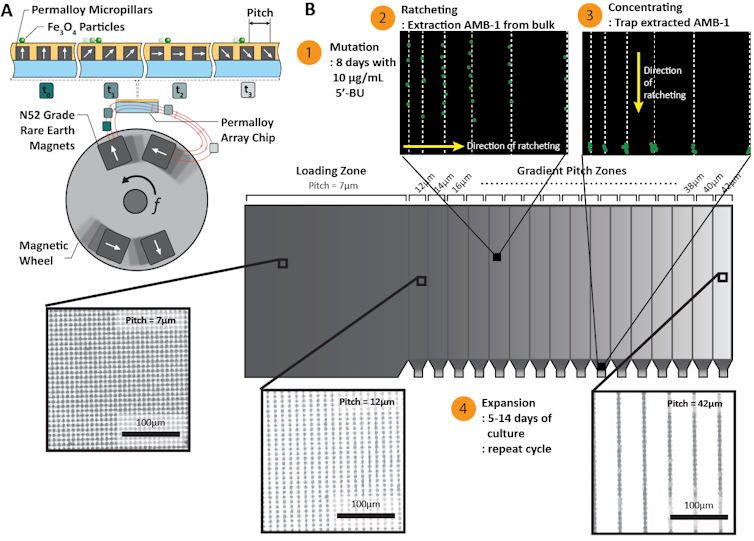
We hope to use our mutation-selection protocol to generate a library of mutants that we can then genetically sequence. Ultimately we want to identify the minimum number of genes we’d need to introduce into a mammalian cell to make it magnetic. Then we could manipulate its activity in deep tissues non-invasively using magnetic fields.
Harnessing their magnetic powers
Magnetotactic bacteria have useful applications even without genetic tweaking.
Researchers have used these bacteria as microrobots for delivering drugs and for removing toxic metals from water. The magnetic nanoparticles they synthesize have also been used in biomedical applications, including targeted drug delivery and killing cancer cells via generated heat, called hyperthermia.
It could be helpful to produce magnetotactic bacteria and magnetosomes on a large scale, particularly the mutants that overproduce magnetic nanoparticles. But scaling up has been difficult so far.
When cultured in large bioreactors, individuals at the top and bottom of the tank experience different amounts of hydrostatic pressure. This can cause them to grow slower and produce fewer nanoparticles. To overcome this problem, I designed a magnetic microfluidic system that can continually sort the bacteria based on their magnetic contents.
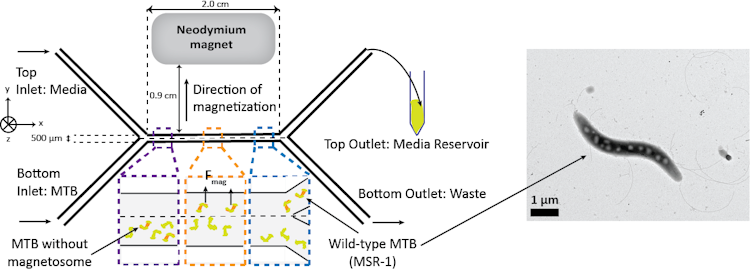
The device consists of a few superfine channels. When magnetotactic bacteria flow in, they experience upward magnetic forces. Only individuals with a user-determined cutoff number of magnetic nanoparticles are collected, while bacteria that failed to reach the mark are disposed of.
This high-throughput cell separation platform allows me to continue culturing only the healthy bacteria which are producing a large number of magnetic nanoparticles. It’s an important step that will help scientists conduct further research in the lab with these intriguing organisms.