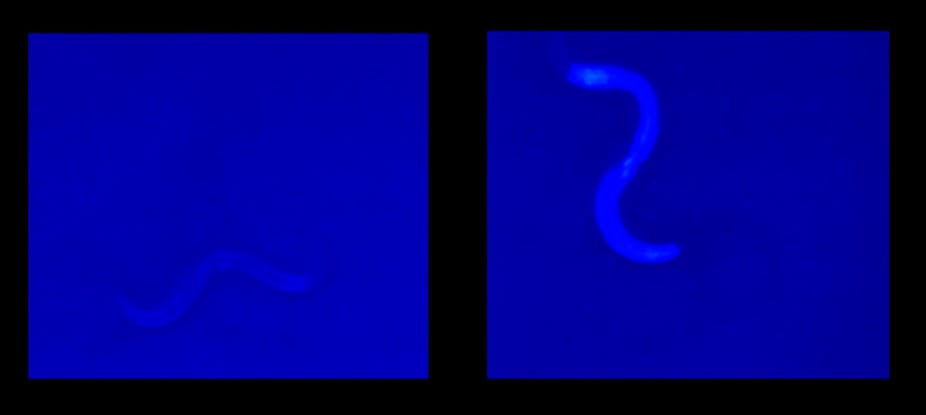
Never has “feeling blue” carried such a sense of finality. A new study has revealed the simple worm (Caenorhabditis elegans) meets its death in a flash of azure. And, according to researchers, the blue light shows that dying may be a coordinated process that could probably be delayed.
Scientists have known for many years that cells can die in two ways. Necrosis occurs when either the cell’s machinery breaks down with age, or exposure to trauma damages the cell beyond repair. Apoptosis is a programmed process triggered on demand, by which even healthy cells can undergo a series of changes resulting in an orderly death.
But when it comes to multicellular organisms dying, such as humans, defining the exact mechanisms becomes more complicated. It cannot simply be explained by a bunch of cells dying independently. How do you define the precise moment when an animal stops being alive and what is the sequence of events leading to death?
To answer that question, David Gems of University College London, who led the study published in the journal PLOS Biology, looked at a simple worm (Caenorhabditis elegans) dying under his microscope. This model organism is easy to study because it is tiny, completely transparent, has a relatively short life and its genes have been studied inside-out.
What Gems and his colleagues found was that under ultraviolet light, the intestine of C. elegans would emit a blue fluorescent light as the worm was about to die. The light, termed “death fluorescence” by the researchers, got more and more intense as the worm’s cells became necrotic. The light reached its maximum intensity at the exact moment the worm died and then quickly faded away.
The finding is remarkable because the blue light was observed both for worms dying from natural causes and for stress-related death, such as under extreme heat or cold. Starting from one end of the worm, the blue light wave would propagate along its intestine until it was dead. This ordered propagation suggested that, among multicellular organisms, death may happen through a series of coordinated “self-destruct” signals.
Gems found that the blue light was caused by the activation of a biological pathway related to calcium signalling, ultimately triggering the fluorescence of small molecules called anthranilic acids (AAs). They found that the spark of blue light was not due to the sudden production of AAs, but because of their release from acidic cellular compartments when the membranes keeping them trapped broke open upon necrosis.
When the team blocked the calcium signalling pathway, the burst could be delayed if a stress was the cause of death. However, no delay could be obtained if it was a case of age-related death. This suggests that age-related death involves more than just calcium signalling.
The work casts doubt on the idea that age-related death is simply a consequences of accumulated damage at the cellular level. It may well be that a coordinated action decides when threshold is met. But the fact that death can now be caught in action under a microscope will help us develop and understand methods to delay it. In which case, seeing the light might be a good thing in the end.