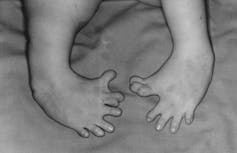
The drug thalidomide has a notorious past. It was used between 1957-62 as a sedative and to treat morning sickness in early pregnancy but a serious side effect resulted in over 10,000 children born with severe birth defects.
How the drug caused these defects, which could affect most parts of the body and includes phocomelia – where the limbs are shortened and missing the long bones with the digits in some cases extruding from the shoulder or the hip – is becoming clearer.
Recent work has shown that the drug is able to inhibit the formation of new blood vessels, induce cell death as well as bind to molecular targets such as cereblon, a protein that alters the function of other molecules to regulate signalling, are all likely responsible for the birth defects the drug caused.
Other uses
Despite the catastrophic actions of the drug when used in pregnant women, thalidomide is now enjoying a renaissance. Thalidomide has been shown to possess a potent ability to inhibit the body’s inflammatory response as well as prevent blood vessel formation in the adult. It is now being used successfully in a wide range of clinical conditions from cancer to leprosy.
Surprisingly, it was just a few years after the thalidomide disaster that the drug was found to be effective for the treatment of Erythema nodosum leprosum (ENL), a complication of leprosy, where painful and inflamed skin lesions occur on the face, arms and legs. Leprosy is seen in tropical and subtropical areas and is particularly prevalent in Brazil and India. Thalidomide is used widely in Brazil to treat this condition, despite the World Health Organization advising against the use of thalidomide for leprosy treatment.
Thalidomide is also very effective for the treatment of multiple myeloma, a cancer of white blood cells. As part of a treatment regime, thalidomide has been shown to prolong the lives of myeloma patients by up to 18 months. The continued study of the drugs ability to prevent new blood vessel formation and its anti-inflammatory capabilities have led to the discovery and marketing of two structurally-related analogs of the drug – Lenalidomide and Pomalidomide – that are more potent than thalidomide and which are used to treat multiple myeloma and complications of this condition.
The mechanism by which these analogs are so effective against multiple myeloma was recently described where the drug binds to Cereblon which has the ability – by altering the function of several other cellular molecules – to selectively kill off myeloma cells.
Thalidomide has also been used to treat Crohns Disease, a nasty inflammatory condition affecting the digestive system with varying success.
It has also been used to treat HIV and complications of HIV, such as Karposi’s sarcoma and several cancers including prostate cancer and brain tumours.
Thalidomide has also been used to treat Behcets disease (an immune system induced vascular condition where vessels are destroyed causing bleeding and tissue damage), as well as graft versus host disorders such as tissue transplants to reduce the chances the host will reject the transplant.
Thalidomide is also effective in the treatment of Hereditary hemorrhagic telangiectasia (HHT) a condition where vessels form abnormally and cause bleeding into organs and from the skin and nose. Thalidomide prevents blood vessel formation and causes stabilisation of the vessels to prevent bleeding.
Unheeded warnings
Thalidomide remains clinically very useful yet long-term use is not recommended due to the ability of the drug to cause peripheral neuropathy, a painful condition that results from damage to the nerves in the body extremities.
Sadly, thalidomide and its analogs still also have the ability to cause birth defects in the developing embryo. To prevent damage being done the US Food and Drug Administration and the EU insist that these drugs should only be taken as part of a carefully regulated and controlled scheme that includes regular pregnancy testing.
Because of this careful regulation, there have been no cases of thalidomide-induced birth defects in Europe or the US since the original disaster.
Tragically, however, the opposite is the case in other parts of the world, notably Brazil where the drug is used widely to treat leprosy complications. Villages in areas endemic with leprosy are often very far from medical help and so a medicine sharing culture exists, and sadly a new generation of thalidomide damaged children have recently been identified in Brazil.
Research into forms of thalidomide that have the clinical benefits but not the side-effects – specifically to prevent its ability to cause birth defect in embryos but also its neurotoxic actions in adults – are underway but this remains a major challenge.
Studying how analogs or structural variants of the drug work and their functions in the body, as well as determining their molecular mechanism/s of action, will shed light on how these drugs act and allow studies into whether the compounds could be useful to treat other conditions including inflammatory conditions such as arthritis.
Such an understanding can also help aid safer and more targeted drug design to combat specific conditions without actions on other tissues or side effects that cause pain.
This is the final article on our thalidomide series. Read the other instalments here.

