Late last year, three distinct and fast-spreading coronavirus variants were observed in the UK, South Africa and Brazil. More recently, variants in India, the US and elsewhere are causing alarm. Does the emergence of these variants portend a protracted battle with the pandemic, or will the virus soon run out of evolutionary room to manoeuvre and settle down as a more benign, endemic pathogen?
Predictions about the evolutionary course of the virus, and specifically changes in virulence, will always be riddled with uncertainty. The vagaries of randomly mutating RNA, chaotic patterns of transmission and expansion, and partially understood forces of natural selection, present challenges to even the most insightful evolutionary soothsayer. Nevertheless, established evolutionary concepts, combined with a wealth of data from the virus itself, can at least provide some pointers.
SARS-CoV-2, the virus that causes COVID-19, jumped into humans from an unidentified animal host, and in doing so entered a new evolutionary space full of obstacles, threats, dead ends, and – very occasionally – opportunity. This space is difficult to imagine and measure. It is annoyingly multidimensional and its boundaries and topographies can be viewed from many esoteric vantages.
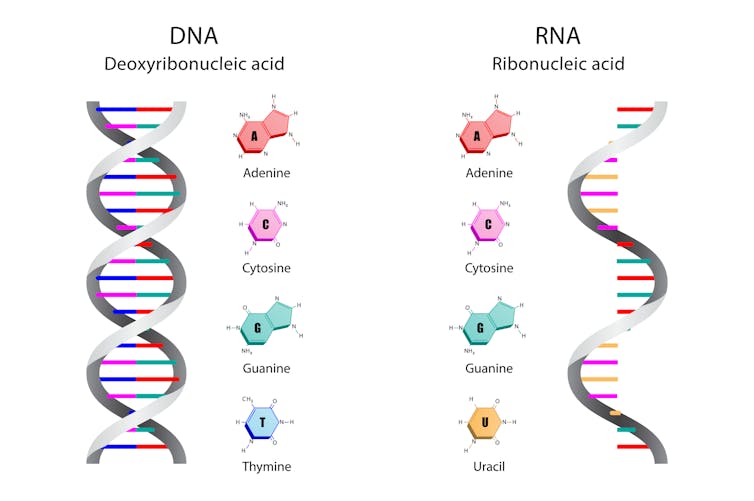
An unsophisticated entry point is to consider the upper limits to genome sequence diversity, or the boundaries of mutational space. Assume the SARS-CoV-2 genome is 30,000 sites long, each of which can be occupied by one of four bases (adenine, cytosine, guanine and uracil). It follows there are over a quintillion (four to the power 30) possible genome sequences, roughly equivalent to the width of the Milky Way in metres.
DNA and RNA both comprise just four bases
But this mathematical limit takes no account of biology and as such is completely unhelpful. Hardly any of these hypothetical genomes would encode a virus that is able to infect and replicate. This basic requirement to maintain the viral machinery in top working order is good news. Evolutionary constraints to maintain viral function will limit how quickly and how well the virus will adapt.
There is more good news. Much of the expert commentary regarding the threat from mutations in the SARS-CoV-2 genome has been relatively sanguine, a reflection of the fact that the vast majority of mutations in the genome have little or no functional consequence. While there may be vast tracts of mutational space that the virus could explore in theory, almost all of this space will be off-limits for a functional virus. And much of the rest will have little relevance to how the virus behaves.
But while mutations are mostly harmless, there remain hidden in deep mutational space, like microscopic dots on microscopic dots, very rare genomic changes that allow the virus to innovate.
How fast is SARS-CoV-2 evolving?
Whole-genome sequencing has provided us with a means to witness the exploration of SARS-CoV-2 through mutational space in exquisite detail. We know that the genome acquires one or two mutations a month on average, which, taking genome size into account, is a rate about four times slower than the flu virus. This was also widely interpreted as good news. The logic here is that a more stable genome provides fewer opportunities for the virus to escape the vaccines or pull off some other genetic trick.
The emergence of the “UK variant” (aka “Kent variant”, B117 or 20I/501Y.V1) was remarkable and sobering. Genome sequence data revealed that it had picked up 23 mutations (a whole years’ worth) seemingly all in one go. Most of these mutations are of little evolutionary relevance, but others are responsible for the increased rate of spread of this variant.
Why and how did this happen? The virus mutates during replication inside a human host. These mutations can change the way the virus interacts with human cells, including the immune system. The UK variant may have emerged during the course of a long-term infection in a single immune-compromised patient. Over many weeks, treatment with convalescent plasma – which is laden with antibodies – could have resulted in natural selection singling out this variant.
Other similar bursts of evolution have been observed. A recently emerged “variant of interest” has arisen in Tanzania that has 34 separate mutations. And examples of recombination, where different SARS-CoV-2 genomes combine to form a hybrid, have also been observed. While these events may be rare, their potential evolutionary significance should not be downplayed. A relatively slow average mutation rate does not automatically correspond to a slow evolutionary rate.
Many are called but few are chosen
The spark for the emergence of the UK variant was probably evolutionary pressure to protect the virus from therapeutic antibodies (convalescent plasma). The most obvious consequence of this was an increased propensity to spread between hosts. This shows that the same mutations can provide the virus with multiple advantages simultaneously, a phenomenon known as “pleiotropy”.
While we may not fully understand the biological mechanisms, we can easily identify candidate mutations from the genome sequence data, because they have emerged over and over again during the course of the pandemic. Although mutation itself is a blind process, natural selection has repeatedly picked these same mutations. This is known as “evolutionary convergence”.
These dozen or so mutations, in various combinations, are the defining feature of all variants. All alter the spike protein – the part of the virus that binds to human cells. Despite the increased caseload and death resulting from these mutations, can we at least take cold comfort from the fact that the total number of such mutations appears to be limited?
Perhaps. But the properties of the virus are probably not determined by single mutations in isolation, but in how several mutations interact. This combinatorial perspective suddenly opens up new zones of potentially fruitful mutational space for the virus.
Appreciating how single rare events can alter the trajectory of the whole pandemic alerts us to the dangers of uncontrolled spread. Just as the chances of winning the lottery increase with more tickets bought, so the likelihood of rare evolutionary events leading to new variants of concern will increase as greater numbers of people are infected.
New variants are no respecter of national borders and, regardless of how difficult it may be to predict evolutionary changes, one thing is clear: from an evolutionary perspective, it is imperative to keep global case numbers as low as possible.