Kidneys are not romanticised like the heart, or held on high like the brain. But these fist-sized organs are integral to our ability to filter waste and excess fluid from our bodies.
And they’re a pain in the midriff when they go wrong.
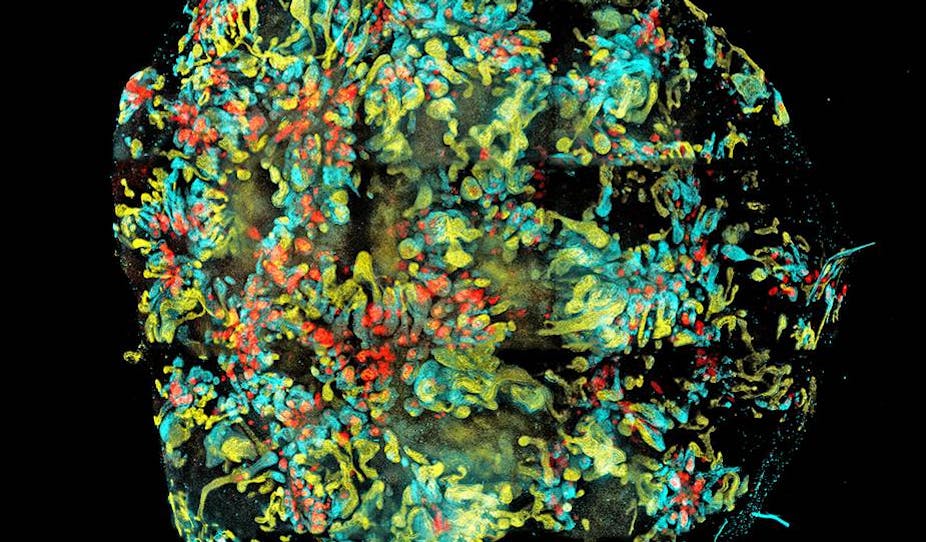
But now it’s possible to grow new kidneys from scratch – at least small ones. This breakthrough in bioengineering was published today in Nature and is a big step forward in regenerative medicine.
One of the paper’s co-authors is Professor Melissa Little, who heads the Kidney Research Laboratory at the Murdoch Childrens Research Institute, and has studied kidney formation for 25 years. She wanted to develop a way of examining how gene mutations create disease.
“We can use this mini kidney model to test drive these mutations, as well as test possible drugs,” she says.
A silent killer
Chronic kidney disease is a growing health problem. One in three Australians are at risk of developing kidney diseases, but the symptoms are so subtle that there’s often no warning.
Many individuals can lose up to 90% of their kidney function before they experience symptoms such as tiredness, puffiness, changes in urine, nausea and blood pressure.
Current treatment programs for kidney disease cost Australia about A$1 billion per year. Primary health care organisations support early detection but that requires people to seek screening from their GP.
Population-wide screening is considered highly expensive and instead is generally targeted to at-risk groups or offered on an individual basis.
So what happens if kidney disease is undetected? Dialysis users sit in a chair or bed for four hours at a time while their blood is pushed out of a catheter in their chest to be cleansed.
To some this represents one of the greatest success stories of modern medicine. For others it’s uncomfortable, boring and potentially fatal. Dialysis can also put a strain on hospital resources with 22,000 people receiving treatment annually.
Kidney transplants are another option but donor rates in Australia are low at only about 16 in every million people. Last year 905 kidney transplants were performed, but for every transplant, another person is waiting and may do so for three to seven years. Even so, making sure the organ is suitable for a person requires both a closely matched blood type and a tissue test to ensure antibodies don’t attack the donor kidney.
Growing a solution

Professor Carol Pollock, from Sydney University, is a medical advisor at Kidney Health Australia and she says the structure of the kidney makes it difficult to grow.
“It’s a very complex organ,” she says. “So to try and have a stem cell that differentiates into more than 20 cell types and functions is much more complex than generating skin cells into heart muscles which have one function.”
Previous studies on growing stem cells into organs generated some of the cell types needed, but didn’t have all of the necessary cell parts. This latest development uses human stem cells and has made a much more complex organ.
The process takes three weeks to get about 20 kidneys smaller than a thumb nail. During this time 10 to 12 cell parts in a dish self-organise into a structure with tubes, blood vessels and the cells in between.
Professor Little says current thought about the process is that the cells move around on their own.
“They rearrange like a room of friends,” she says. “They respond to what’s around them to make a structure.”
We are not yet at the stage where we can grow full size human kidneys, but the potential for using stem cells to bioengineer functional organs is an exciting goal.
“Assume you have kidney disease late in life rather than inherited disease, we could theoretically take your skin and develop a new kidney,” Professor Little says.
For now, these mini kidney models can be used to test drugs, treatments and research the development of disease. It’s one small step for humans, but one giant leap for regenerative medicine.