SAVING THE OZONE: Part two in our series exploring on the Montreal Protocol on Substances that Deplete the Ozone Layer – dubbed “the world’s most successful environmental agreement”. Yesterday’s article looked at why the protocol was a success. Today, what is the ozone hole?
Ozone is fundamentally important for life on Earth. It forms a natural layer in the stratosphere which acts as a sunscreen to filter out harmful ultraviolet radiation.
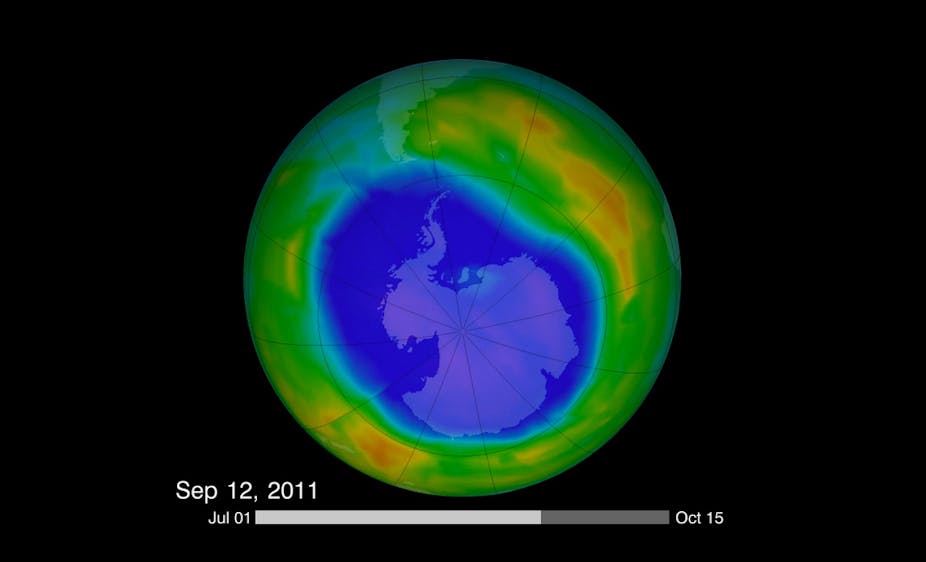
Human activities have had a dramatic impact on the health of the ozone layer, and we are still witnessing these effects most significantly at high latitudes. For more than three decades Antarctica has experienced the most severe depletion of stratospheric ozone in what is commonly referred to as the Antarctic “ozone hole”.
So how does the ozone hole form and what is the current outlook for ozone?
Chemical ingredients
The most potent man-made ozone depleting substances (ODS) are those that are halogenated – that is, they contain chlorine and bromine.
Halogenated ODS are effectively inert in the lower atmosphere. The overturning motions associated with warming and cooling of airmasses in the lower atmosphere allows some of the ODS to be transported into the stratosphere. The substances are then broken down by ultraviolet light and form stable halogenated “reservoir” compounds, which by themselves do not strongly affect ozone.
The main problem arises when the reservoir compounds are further altered to create halogenated “reactive” radicals. This is done through processes that largely take place on the surfaces of tiny stratospheric aerosol particles. These reactions make the by-products of the ODS more potent for ozone destruction.
The amount of particles taking part in this process is significantly enhanced in Antarctica over winter. This is because of the presence of Polar Stratospheric Clouds (PSCs) in the lower stratosphere.
Special Antarctic conditions
PSCs are made up of tiny particles made up primarily of nitric acid and water vapour, and require a temperature of around -80°C or colder to form. In winter, a vortex of air forms in the stratosphere over Antarctica. Within the vortex, air circulates with minimal mixing with the rest of the atmosphere. This drives strong cooling of the lower stratosphere and creates the conditions favourable for PSC formation.
When spring comes, the halogens released by the PSCs are broken down by sunlight. The resulting free chlorine and bromine atoms destroy ozone before being cycled back into halogen radicals. The presence of sunlight begins the cycle again.
Overall, the reaction cycle destroys many ozone molecules for each chlorine and bromine atom present. PSC formation enhances the cycle as it removes certain nitrogen compounds from the atmosphere. These compounds would otherwise reduce the overall amount of halogenated radicals present.
Life cycle of the hole
Ozone destruction peaks in early spring and ends by late spring or early summer. The warming atmosphere evaporates the PSCs thereby inhibiting the cycle. It breaks down the vortex, resulting in the dispersal of ozone-poor air to mid-latitudes and allowing ozone-rich air from outside the vortex to flow over Antarctica.
Ozone destruction is related to the amount of chlorine and bromine present in the polar stratosphere. This is influenced by the amount of chemical processing by PSCs, along with the size, stability and temperature of the vortex.
The Arctic and the rest of the globe
Ozone destruction also occurs in the Arctic, although with less severity and consistency than Antarctica. The Arctic winter stratosphere is less cold and stable than that of Antarctica, producing lesser amounts of the destructive halogenated radicals.
Ozone depletion reactions triggered by the breakdown of ODS in the stratosphere take place around the globe, although with far less effectiveness than in the Antarctic.
The Antarctic ozone hole: looking back and forward
The Antarctic ozone hole was first clearly apparent around 1980, although signs of ozone decline at specific Antarctic sites have been traced back to the 1970s and possibly earlier.
The various metrics that assess the size and severity of the ozone hole show that the Antarctic hole grew through the 1980s and generally levelled-off in size during mid-1990s. Dominant factors have been the size and strength of the polar vortex and temperature of the lower stratosphere in the time since.
Year-to-year size variations in the ozone hole are driven largely by meteorological variability rather than changes in ODS. Years where the polar vortex was large, stable and cold, such as 2000 and 2006, are associated with the most extensive ozone loss. An unusual year was 2002, when the polar vortex broke down in an unprecedented and dramatic way after a winter of relatively mild stratospheric temperatures and disturbed circulation. In that year, the severity of ozone loss was lower, similar to the mid-1980s.
In recent years there is increasing evidence that the declining level of ODS are playing a mitigating role in the overall size of ozone hole.
Clearer evidence of a reversal in the severity of Antarctic ozone hole should emerge over the next decade. The trend produced by the decline in ODS will become more apparent against year-by-year variability from meteorological processes. The current expectation is that pre-1980 levels of ozone in the Antarctic region will return around the middle of this century.
Tomorrow: how does the ozone layer protect Earth from radiation?
Read more on the Montreal Protocol’s 25th anniversary.