Gold has extraordinary properties. It can be used to make jewelry, but also to fight cancer. Several clinical trials are currently underway in the United States where patients are being treated with gold nanoparticles.
In its natural state, gold is a yellow, chemically inert, non-corrodible metal, making it a noble material that does not degrade over time. These properties, along with the ease with which it can be shaped, have made it the favourite metal of jewellers.
On a nano-metric scale – that is, at a millionth of a meter – gold has other remarkable properties. On this scale, gold particles take on various colours according to their shape and size. This property has been used since ancient times to colour glass and earthenware – giving them, for example, an intense ruby hue. When light is shone on gold nanoparticles, the metal’s conduction electrons are excited by the light wave and begin to oscillate. This oscillation is particularly intense for a given colour in the light spectrum. This is called resonance.


By changing the shape or size of the nanoparticles, it is possible to choose the resonance frequency that has the strongest interaction with light. The nanoparticles then behave like tiny, highly effective antennae, and although they are extremely small and highly diluted they can give vibrant colours to stained glass.
One incidental consequence of this intense interaction with light is that nanoparticles heat up. This remarkable property is the reason behind their use in new cancer therapies. The idea is to destroy the tumours with photothermia – in other words, to locally heat up tumours “decorated” with gold nanoparticles by exposing them to light.
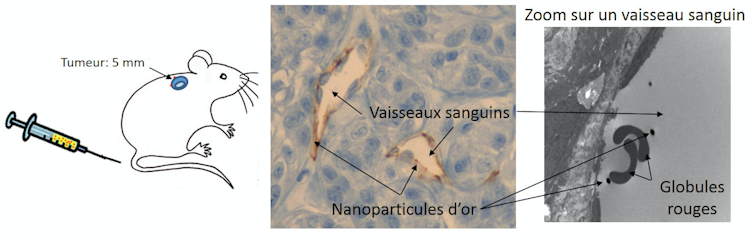
Patients being treated this way are first injected with gold nanoparticles into their bloodstream through an IV. Since gold is biocompatible, it presents no apparent danger to health in the concentrations used in therapy, as borne out by our studies in mice. However, not all questions have been resolved concerning these new applications. Gold nanoparticles go undetected by the body’s immune defence system. Their nanometric scale means they are generally one hundred times smaller than cells, allowing them to move freely through the blood system and enter the tumour.
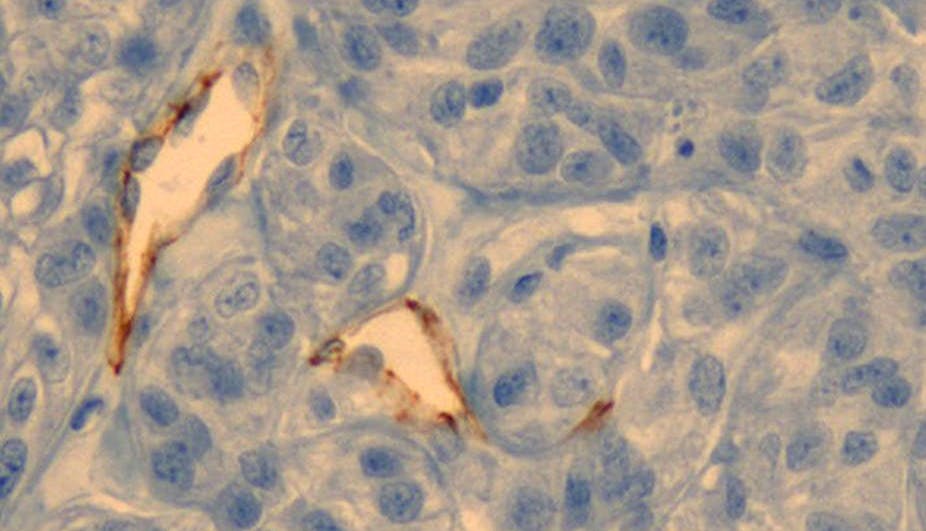
They must then be concentrated inside the tumours, many of which are highly vascularized – they naturally acquire a network of blood vessels allowing them to grow. Using this pathway, the nanoparticles easily accumulate inside the tumour. The altered structure of the blood vessels in the tumour area makes them more permeable, facilitating a high retention of nanoparticles.
Tumours “decorated” with gold nanoparticles are then exposed to light, in order for them to heat up and be destroyed. At this stage, the challenge is twofold. While the light must penetrate the body and reach the tumour, healthy tissue must not be heated. The choice of frequency is therefore vital. Nanoparticles must be lit up at their resonance frequency, but it is just as essential that the tissues without nanoparticles not absorb the light.
While our bodies absorb light in the visible part of the light spectrum (that is, all the colours of the rainbow), this is not the case in the near infrared. We can see this by simply placing a hand over an intense white light. Only the colour red, on the edge of infrared, can move through the flesh of the hand.
This range of the spectrum in the near infrared is often called the “therapeutic window” – the range that can be used in medical treatment. In the visible spectrum, light is mainly absorbed by hemoglobin, while light further into infrared range is absorbed by the water contained in our bodies.
Nanoparticles with specific shapes
By playing with the shape of nanoparticles, it is possible to adjust their resonance so as to target the near infrared therapeutic window. This is carried out, for example, for nanoparticles with a silica core and a gold shell, for gold nanorods, or for nano-cages shaped like porous cubes. Preclinical studies (in animals) have enabled us to test the safety and effectiveness of various shapes of nanoparticles.
In the therapeutic spectrum, light goes through our bodies, but our bodies are not totally transparent to it. The light that comes out the other side is still highly diffused by the body’s tissues. For instance, one cannot see bones in this way, as you would with an X-ray. It is also very difficult to focus a beam of light on a tumour from outside the body, since the light must travel through healthy tissue to reach it.
It is therefore usual (in animal studies) to light up tumours more closely, by inserting a needle through the skin, attached to an optical fibre linked to an infrared laser. The light is then far more intense in the relevant area.
Studies underway on head and neck cancers
Under the light, the gold nanoparticles heat up and “cook” the tumour, thus destroying nearby cancerous cells. Extensive studies have been carried out in animal models on cancers in the brain, prostate and pancreas, for example. Clinical trials are also underway in the United States in patients affected by treatment-resistant head- and neck cancers, and lung- and prostate cancers using AuroLase therapy (Nanospectra Bioscience).
Alternatively, nanoparticles can be used not as a direct weapon against the tumour but as a means of transport (called a vector) to deliver molecules – drugs, for instance – to their destination. This technique requires less heating. The use of vectors should reduce the toxicity of treatments by better targeting cancerous cells.
The Trojan horse strategy
It is possible to increase the number of gold nanoparticles entering a tumour, above and beyond the effect of simple passive accumulation. They perform better when covered with molecules (antibodies) that specifically attach to cancerous cells, which they recognize through the proteins present on the cell membrane. Other alternative techniques adopt a “Trojan horse” strategy. These use a kind of white blood cell, called microphages, filled with gold nanoparticles in order to penetrate more deeply into the tumour.
Gold nanoparticle photothermia is a promising new therapy in cancer treatment. It has begun to be used experimentally in patients with certain specific cancers, but much research is still necessary before it can be adopted more widely. In the future, the technique will have to target the tumour more effectively and exclusively. With thriving research, this therapy should be available, alongside existing treatments like radiotherapy and chemotherapy, in a few years’ time.

Created in 2007, the Axa Research Fund supports more than 500 projects worldwide led by researchers of 51 nationalities. Discover the work of Emmanuel Fort and his Axa ESPCI chair in biomedical imaging as part of the Axa Research Fund.
Translated from the French by Alice Heathwood for Fast for Word