Our understanding of the natural world is now so great we can manipulate the DNA blueprints for any living thing on Earth. We can replace genes for traits we don’t like with others we prefer and even add genes that don’t occur naturally in an organism. Over the last few years, scientists have developed several methods for editing genes in this way and excitement over one in particular, the CRISPR-Cas9 system, has reached fever pitch.
We have also developed a way to introduce these gene changes to an entire population of a species. This “gene drive” process has most recently been used to alter the DNA of small groups of mosquitoes so that they no longer carry the malaria parasite, raising the possibility of eliminating the disease altogether. But meddling with nature in this way carries huge implications that need careful consideration.
Gene editing
Gene-editing techniques involving cutting genes at specific sites in the DNA of an embryo in order to disrupt those genes’ function or insert other genes. For instance, the CRISPR-Cas9 system uses enzymes that can cut specific gene sequences from DNA, guided by a similar molecule known as RNA. Natural gene repair mechanisms then kick in and can be used to disrupt the function of the original gene or replace it with a completely different one.
CRISPR systems actually aren’t new – they have existed in nature for millions of years. Bacteria use them to fend off viral infections by adding part of the virus’s DNA to their own. So why all the fuss? CRISPR-Cas9 makes artifical gene-editing much easier and cheaper, enabling scientist to target specific bits of DNA. By comparison, another method known as TALENS requires the construction of complex proteins. As a result, CRISPR gene-editing is heralding advances in biomedicine such as cancer treatments and protecting individuals from infections
But there are other ways gene-editing has the potential to help in the fight against infectious diseases. Very recently, CRISPR methods have been used to make mosquitoes resistant to malaria infections and coupled with a “chain reaction” to drive this gene modification (the resistance to malaria parasite) through the population.
Gene drive
This process is referred to as a “gene drive”, and again is not new: nature spreads evolutionary changes through a population all the time. It doesn’t mean changing the DNA of all living individuals in a population. Instead it’s about ensuring a specific genotype (a certain version of a gene) is passed on to the descendants of modified individuals.
A sexually reproducing organism usually has a 50% chance of inheriting a specific genotype from one of its parents. Using a gene drive can bias the inheritance pattern to increase that chance to nearly 100%, ensuring almost all descendants possess the genotype. As those descendants mate and produce their own offspring, the proportion of organisms with the genotype increases until it can be found in the entire population.
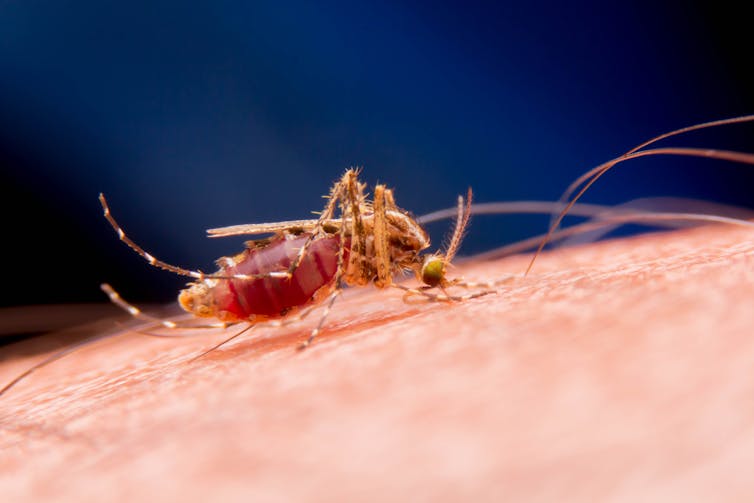
The idea that you can “replace” a population’s genotype is particularly appealing when that population is responsible for spreading disease, as mosquitoes are with malaria. Malaria is preventable and curable but still kills over 400,000 people each year.
The potential for using a gene drive to engineer insects (particularly mosquitoes) was discovered in the 1960s. But the advent of CRISPR’s cheap and easy gene-editing puts this research onto a whole new footing. Researchers at the University of California, Irvine, recently published a proof-of-princple study demonstrating the techniques can alter a population of the main type of mosquito that carries malaria in urban India, Anopheles stephensi.
Putting into practice
The longer term aim, in this instance, might be to release a persistent, modified mosquito into the environment to assist in the control a public health problem. This would be an area-wide release programme to compliment existing control interventions that would require case-by-case assessment of all the cost and benefits. For example, mathematical modelling would be needed to work out how many modified mosquitoes to release, how long it would take for the mosquito population to be clearly affected and how long it would take to impact public health.
One obstacle to the practical use of gene-drives is the need for relevant regulations, or at least the application of existing laws on genetic modifications. Gene-drive technologies are still some way off from the necessary environmental risk assessments for field trials and releases that would sufficiently scrutinise the risks to the environment and/or human health. These sorts of CRISPR-based modifications might even need a whole new set of regulatory structures that require a fuller debate about novel biotechnological advances.
Rapidly targeting genome modifications has the power to advance many aspects of basic and translational biomedical sciences. The potential benefits to reducing the impact of infectious disease and genetic disorders, including cancers, and improving the way the immune system works are huge. But the technology isn’t without pitfalls.
CRISPR systems rely on a guide molecule to make sure the DNA sequence is cut in exactly the right place. Getting this wrong will probably cause damage to non-target genes that could harm the organism. And just because we can edit the DNA within a species doesn’t mean we should. We need strong leadership at all levels – ethical, scientific, political – and appropriate regulations to ensure these new technologies can prosper without unintended consequences.

