Last year Japanese scientists announced they had for the first time extracted gas from offshore deposits of methane hydrate, an ice-like substance made of natural gas (methane) trapped inside water crystals.
For resource-poor Japan, which must import most of its fossil fuel supply, this could be a route to much-needed energy security, and was hailed as major breakthrough. And with potentially huge reserves of methane hydrates found in deposits around the world, depending on the technology and the need, methane hydrate could be the next important “unconventional” fossil fuel to arrive after shale gas.
Methane hydrate deposits form and remain stable only under high pressure or low temperatures, such as in areas of permafrost, or beneath the pressure of hundreds of metres of water at the bottom of the ocean. The methane that it contains is usually formed by the work of tiny microbes that consume organic material buried in seafloor sediments. Sometimes the methane has a deeper origin, from the breakdown of organic material cooked at the high temperatures that occur several kilometres down in the Earth’s crust, in a similar process to the formation of oil. Once formed, it slowly percolates up through the rock towards the surface.
Because there is a generous supply of organic material to regions of the deep seafloor close to the continents, these regions are believed to contain very large amounts of methane hydrate.
How much do we have?
It’s very difficult to determine an accurate estimate for the actual amount of methane hydrate lying in the oceans. The hydrate is commonly present in a distributed form, either occupying cracks in the sedimentary rocks beneath the seabed, or filling parts of the microscopic pore space within the rock. Hydrate may fill only a few percent of the available space.
Methane hydrate’s physical properties are very different from the salty water that normally fills these spaces. We can estimate some of these properties, such as sound speed or electrical resistivity, by conducting geophysical surveys. However, our surveying methods normally average these properties over volumes that are tens to hundreds of metres across, and on this scale the effect of the hydrate can be quite subtle and hard to recognise.
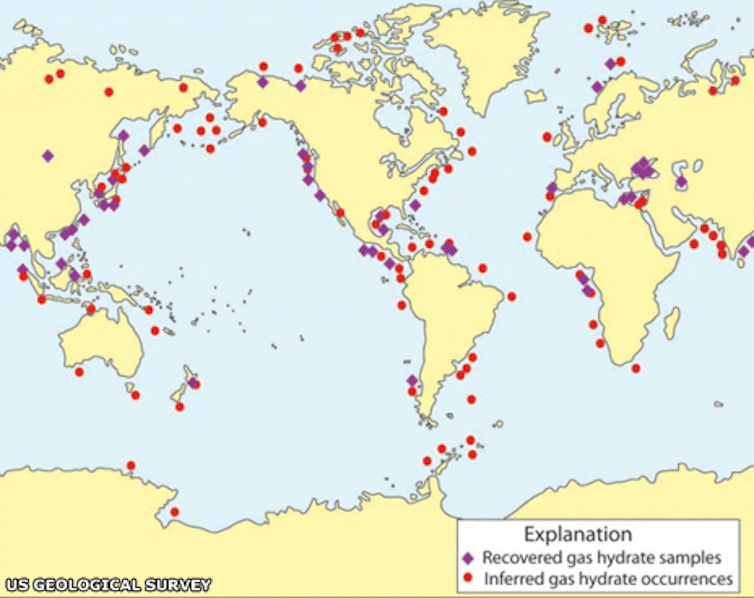
Only a tiny fraction of the seafloor has been surveyed by these methods, so the most reliable estimates of global methane hydrate resources come from computer models. Based on estimates of the amount of organic matter present in seafloor sediments, these models make predictions of how much methane might be generated and converted to methane hydrates. When hydrates were first discovered in the oceans in the 1970s, it was thought that they might be the dominant form of organic carbon on Earth, and a much larger resource than conventional fossil fuels such as coal, oil and gas. But as surveying techniques and our understanding of how methane hydrates form has improved, more recent estimates have steadily declined to below the total amount of fossil fuels. Most recent estimates are around 1,000 gigatonnes or less of carbon bound in methane.
Nevertheless, hydrates probably still constitute a substantial reserve of natural gas and their resource potential has led to several nations, including the US, Japan, China, India, South Korea and Taiwan, launching large-scale hydrate exploration programmes.
How can we use it?
Geophysical surveys are effective at estimating hydrate volumes in those areas where it is more concentrated, particularly when complemented by direct sampling in boreholes. A tell-tale sign of the presence of hydrate is the so-called “bottom-simulating reflector” (BSR). Because temperature increases with depth beneath the seafloor, at a certain depth the hydrate becomes unstable, and below this depth the methane is present in gaseous form and affects sound passing through it differently. This boundary is often a strong reflector of sound waves that roughly parallels the seabed and crosses geological structures, so it is a distinctive feature in images constructed from seismic surveys of the seabed. Exploration effort tends to be focused in areas where BSRs have been detected.
Even once an accumulation of methane hydrate has been found, putting it to use an energy resource is a considerable challenge. Other than in permafrost regions, which present their own challenging, remote and hostile environment, hydrate is present only in deep water where building structures on the seabed is a significant engineering challenge.
Since it commonly lies beneath many metres of rock, using straightforward mining is not generally practical, and exploiting the gas requires removing it from the solid hydrate. This process requires either an increase in temperature or a reduction in pressure, both of which in turn require energy to achieve.
Sometimes nature does the work for us and ocean warming triggers the release of gas, as may be happening near the Arctic archipelago of Svalbard. But such natural release tends to be too widely distributed to be collected easily. So the technological challenge that Japan and others face is to extract the gas and transport it to somewhere it can be used. On top of that already substantial scientific and engineering task, ideally some way of collecting and storing the resultant carbon emissions from burning the gas as energy and putting them into permanent storage is required, perhaps back under the sea floor.
For methane hydrates to be worth exploiting, all of this needs to be possible without using more energy than can be extracted from the methane, else the entire operation cannot be financially viable. And while surveying techniques aimed at locating methane hydrate deposits have improved, we are still – despite Japan’s efforts – some way from making methane hydrates pay for themselves.

