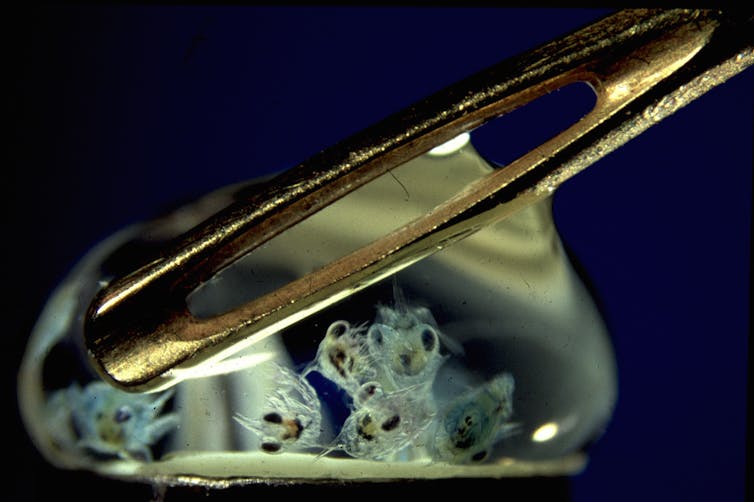
Many people who love the oceans never realize that a single drop of seawater is teeming with plankton, which means “drifters” in Greek. These organisms, which typically range in size from a pinhead down to the tip of a pin, spend their lives drifting with currents and form the base of ocean food chains.
Most larger marine organisms, from corals to crabs to fish, also begin life as tiny drifters. Females release eggs, or larvae, which join plankton and spend days to weeks adrift. Only larvae that arrive at the right habitats, at the right stage in their life cycles, will grow into a new generation. The rest will perish.
Scientists have long assumed that larvae are at the mercy of ocean currents. We cannot track such tiny creatures for weeks and months as ocean currents carry them over long distances, but many of us have wondered where they go and how they get there.
Thousands of miles and decades apart, members of our team independently wrestled with this puzzle. Studying land crabs on Bermuda, Tom and Donna Wolcott of North Carolina State University wondered why larvae of one species seemed to return to the islands every year from the surrounding ocean, but those of another species did not. On the California coast, Steven Morgan at the University of California at Davis documented larval behaviors of shore-dwelling species that apparently enabled them to avoid being swept offshore by strong currents. And Morgan’s colleague John Largier explored the pattern of currents in which the larvae drifted.
We needed a new tool to address these issues. The Wolcotts reasoned that they could not put transmitters on individual larvae, but they could design something that behaved like a larva. If it could be tracked, following a group of them would reveal the path of a cloud of larvae.
Over three decades they developed a robot “larval mimic” that senses its environment, mimics the vertical swimming responses of larvae and relays its location. Following a cluster of these robots would show where larvae would be carried by currents, depending on the depths the larvae chose.
This research is providing insights into how evolution has “tuned” subtle changes in larval swimming behaviors to maximize the chances that larvae drifting in the ocean will beat the enormous odds against them and arrive at suitable habitat for settlement.
Following the currents – or not
The strong and persistent currents off the northern California coast provide an excellent opportunity to test the effectiveness of larval behaviors. Upwelling occurs along coastlines that run north-south along the western margins of continents, where prevailing winds combine with the rotation of the Earth to drive surface waters away from shore. This process draws deep, cold, nutrient-rich waters to the surface, supporting some of the most productive fishing grounds in the world.
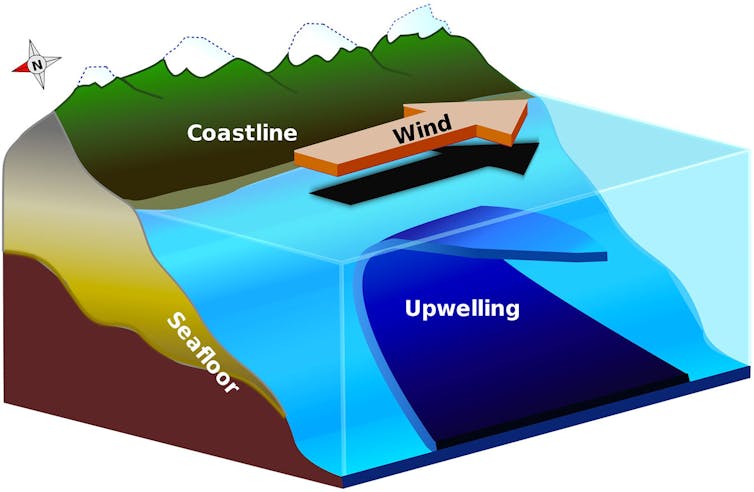
Scientists have long assumed that during upwelling, larvae developing near the surface were carried away from shore and lost at sea. But when Morgan and colleagues towed plankton nets at discrete depths throughout the water column and across the continental shelf of northern California, they found that larvae of most species remained within three kilometers of the shore where they originated.
How do such tiny organisms control their positions in the water? It has been known for decades that even though larvae cannot swim against currents, they can swim vertically from one layer of water to another moving in a different direction. The evolution of vertical swimming behaviors could help larvae to reliably stay close to shore, or to range different distances across the continental shelf.
By regulating their depth, larvae appear to have a surprising amount of control over how far from home they are transported. Many marine populations need this ability for successful reproduction. It also determines which populations exchange larvae sufficiently to remain a single genetically similar population along the coastline.
Knowing that larvae can regulate their movements in this way could affect how we design and evaluate networks of marine protected areas to conserve ecosystems and populations of economically and ecologically important species. It also is relevant for tracking the spread of invasive species, and for analyzing how marine species adapt to a warming planet by shifting their ranges north or south along the coast.
A robot that mimics larvae
Tom and Donna Wolcott named their robot the Autonomous Behaving Lagrangian Explorer, or ABLE – autonomous because it is deployed without a tether, and Lagrangian because it is carried along with the water parcel in which it is embedded. The Lagrangian approach to studying currents consists of following the movement of a parcel of water, typically with some sort of “drifter” – in this case, a drifting robot.
But the ABLE is not entirely passive: It mimics larval behavior by monitoring time and conditions around it, including depth, temperature, light, salinity and vertical water motion every 10 seconds. Then it calculates a new “target depth” appropriate for that larva and swims toward it by subtly adjusting its buoyancy. It can log up to 16 megabytes of environmental data from its sensors and upload them after the “bot” is recovered from the water.
When the Wolcotts snorkeled with several of the initial ABLEs in the Bahamas in 1994, they could see them swimming up and down as programmed. The ABLEs could also be tracked by following signals from their ultrasonic pingers – until drowned out by the din of snapping shrimp.
Next the Wolcotts created ABLEs to simulate mobile phytoplankton for research by North Carolina State University marine scientist Dan Kamykowsky on transport of “red tides,” which are dense blooms of toxic algae. The Wolcotts added a light sensor so that ABLE could calculate photosynthesis and respiration. The ABLEs could then mimic the movements of the algal cells, moving upward to meet their needs for light and photosynthesis and deeper to find essential nutrients.
These ABLEs also carried redundant tracking hardware: GPS receivers to get position fixes, VHF radio beacons to transmit those positions and LED beacons so that researchers could retrieve them from the water after dark. Bluetooth wireless connectivity enabled the bots to talk to computers while their pressure housings were sealed. This version successfully emulated red tide algae during experiments in the Gulf of Mexico.
The newest ABLEs carry a transmitter to relay the latest GPS fixes via satellites to our computers and smartphones, and a sensor to keep the robots from getting stuck on the sea bottom. Running all of this gear and simulating larval swimming behaviors requires a program that contains roughly 3,000 lines of code.
Cracking the black box
Steven Morgan first saw the Wolcotts demonstrate an ABLE prototype in a hotel pool at a 1996 conference. In 2015, after two more decades of “ABLE evolution” and field testing, we conducted the first experiments using them to understand how larvae were transported in currents near the University of California at Davis’s Bodega Marine Laboratory in northern California.

Circulation is more complex around headlands and bays than along straight coastlines, which makes it especially hard to predict where larvae go. Based on prior study of larval behavior and currents, Morgan and Largier predicted that during upwelling, larvae in surface waters would be transported far to the south, while larvae in bottom waters would stay put. They also expected larvae that remain in bottom waters during the day, rising to the surface to forage at night after winds subside, would stay put.
To test this hypothesis, we repeatedly released ABLEs programmed to simulate larvae with different vertical swimming behaviors and tracked them for 24 hours. Their movements demonstrated that larvae that remained near the surface would be transported up to 8 kilometers southward within 24 hours. In strong contrast, ABLEs that stayed deep in the water column remained near where they were released. ABLEs that migrated to the surface at night did not travel much farther than the ones that remained deep, moving less than 2 kilometers.
From these experiments, it is evident that vertical positioning in currents, even if cued only by the clock, has profound effects on larval movements. ABLE’s ability to respond to environmental cues, and its battery life of over two weeks, also will enable us to examine how changes in depth preferences affect transport of larvae during the course of development.
Larvae of many species occur near the surface in offshore-flowing currents early in their development and are carried away from shore. Older larvae descend into deep shoreward-flowing currents and return onshore to habitats in which they can metamorphose and become adults. In effect, they use currents like conveyor belts to travel out and back across the continental shelf.
Now we are deploying ABLEs with different behaviors to collect the first experimental evidence of how patterns of vertical migration by larvae determine how far offshore and alongshore they travel. This information will help us crack the “black box” and reveal where larvae of different species go.
We also plan to deploy ABLEs in different environments, such as estuaries. Eventually we plan to return to Bermuda or another isolated island to answer the Wolcotts’ original question: How do larvae find their way back to the shores of a speck in the middle of a vast ocean?

