The book “My Big Quantum Meccano”, published by Flammarion, describes 11 of the most amazing discoveries in quantum physics.
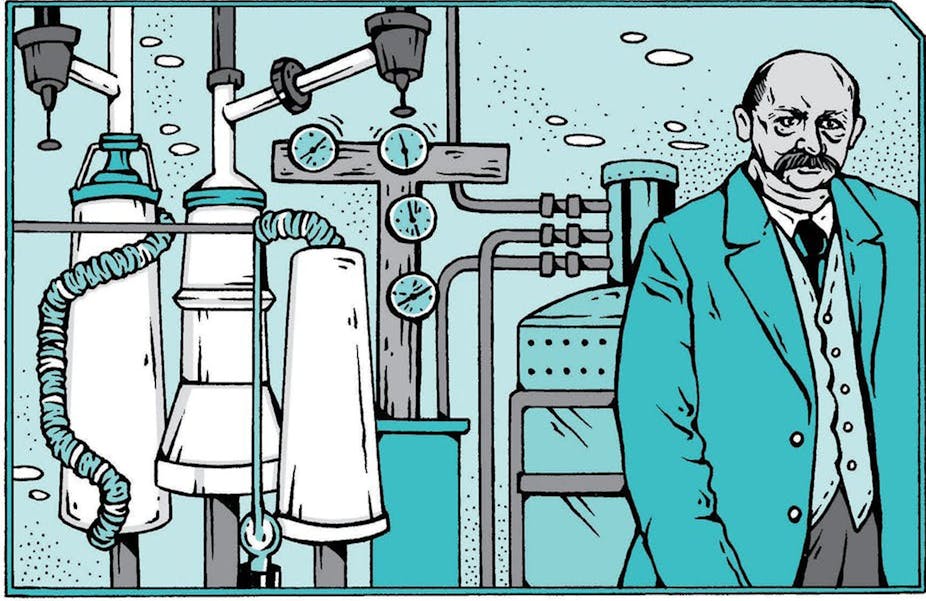
The setting for this story is Leiden University in the Netherlands. As you step into the laboratory, you’re struck by the size of the pumps and their deafening noise – the floor vibrates. The three physicists in the room are busy. Gerrit Flim, the team engineer, is watching over a big white frosted tank with a large number of tubes and wires coming out of it. Cornelis Dorsman is helping him. Behind them is a third man, lab director Heike Kamerlingh Onnes. The 57-year old is bald, sports a bold mustache and wears a lab coat over his spotless suit. He is overseeing operations and making notes meticulously.
On his right, a man’s voice coming from a tube calls out: “Zero, zero, still zero!” The tube comes from an adjacent room, where we find physicist Gilles Holst, barely 25 years old, sitting opposite an electric machine. His eyes are fixed on a spot of light on the wall and he’s yelling himself hoarse, quite uselessly at this point, because what is happening in this lab is truly beyond comprehension.
Let’s take a closer look at the equipment, starting with the white tank first. This is a glass cryostat, a sort of very large thermos, keeping its contents perfectly insulated from room temperature. That’s hardly surprising, given that the temperature inside the cryostat at that precise moment is the lowest ever achieved: -269°C, in other words, only four degrees away from absolute zero, which is almost 100 times as cold as the laboratory and 50 times as cold as anywhere on Earth, even at the South Pole. Kamerlingh Onnes had set this world record three years previously, by making helium liquid […] a feat which earned him the nickname of “the gentleman of absolute zero”.
Liquid helium to test metal conductivity
Let’s go back to that Saturday in April 1911, when Kamerlingh Onnes now decides he wants to test the electrical properties of metal. The question is a simple one: is a metal more or less conductive when it is subjected to extremely low temperatures? Electrons do the conducting and behave like a kind of electric liquid, flowing among atoms. To analyze their behavior, you just need to measure their electrical resistance. The lower the resistance, the easier it is for the current to flow. The resistance of an insulating material, such as plastic, can be a billion times higher than that of aluminum or mercury. That sets Kamerlingh Onnes thinking: if you cooled metal, wouldn’t electrons also end up being frozen and immobilized and therefore unable to conduct the current? Would metal become insulating at the temperature of helium? Would resistance increase to the point of becoming quasi-infinite at low temperatures?
“Test gold”
Kamerlingh Onnes prefers experiments to predictions, in keeping with the motto displayed at the entrance of his laboratory: “Knowledge through measurement”. He chooses mercury, a metal that is liquid at ambient temperature. He does this because it can be made pure by a distillation process, and measuring wires can be immersed into it, with no welding required. To measure resistance, the team uses the most sophisticated technique available in 1911: a Wheatston bridge and a mirror galvanometer. The “bridge” is an electrical circuit that compares the resistance of mercury with other known resistances. The galvanometer indicates the value of the resistance being measured, thanks to a light beam deflected by a mirror.
Holst is next door, watching the resulting spot of light and yelling out its position through the tube. He’s being kept some distance away, to make sure that vibrations from the pump don’t interfere with the galvanometer or the light beam. There is no IT, oscilloscope or multimeter here, all the work is being done by hand and by sight.

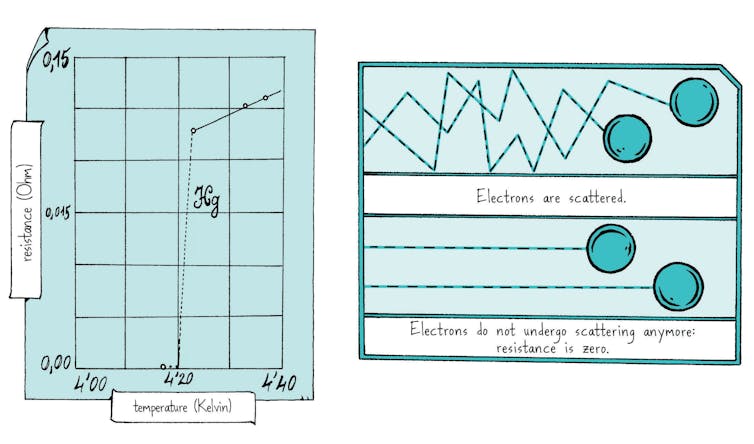
The unbelieving Holst stubbornly calls “Zero!” The resistance of mercury is zero, that’s what the galvanometer is saying. What’s happening seems to be impossible, it goes against all predictions. The physicists’ first thought is that there might be a short circuit. If the measuring wires are touching one another, the current will flow directly from one to the other, exactly as if the mercury weren’t there, and it will look like there’s no resistance at all.
They decide to warm the sample to check the connections, which brings the second big surprise of the day: as soon as the temperature again rises above -269°C, the spot of light suddenly starts to shift. Resistance reappears, which means that this was no short-circuit. The massive drop in resistance can be both reversed and reproduced and can always be observed at precisely -269°C. Kamerlingh Onnes jots down in his notebook: “Mercury at zero” and straight after, adds: “Test gold”.
Perpetual movement?
Kamerlingh Onnes later names this sudden and unexpected movement “superconductivity”. He comes up with this name on day he receives the Nobel Prize, two years later. Superconductivity describes the ability certain metals have to conduct electricity perfectly below a certain very precise temperature. Superconductivity is later measured in a number of metals, including aluminum, tin and lead.
A year later, Kamerlingh Onnes carries out an even stranger experiment. He builds a tin ring, which he connects to a battery to create an electrical current, he then cools the ring to make it superconducting and disconnects the battery. If electrical resistance really is equal to zero, then nothing can resist the current, which has to stay trapped in the ring, circling it forever.
Kamerlingh Onnes waits a little, then places a compass close to the ring. Amazingly, the needle swings round. This proves that the electric current is still flowing in the ring, creating a magnetic field. Kamerlingh Onnes confirms the surprising properties of superconductors, this time by showing that electric currents can be trapped forever!
People sometimes say that Kamerlingh Onnes was really lucky to come across this phenomenon. They even talk of serendipity, a happy coincidence, whereas it was nothing of the sort! It’s true he didn’t expect to find what he did, but things that are unexpected doesn’t necessarily happen randomly. This particular discovery was the result of more than ten years’ concerted effort, involving a great team of physicists, engineers and exceptional technicians who, right from the start, wanted to probe matter close to absolute zero and worked tirelessly to achieve that.
It took 45 years to understand the reasons behind this strange phenomenon. Electrons in metals behave like little quantum waves. At very low temperatures, thanks to the atoms’ vibrations, they combine, first in twos and then all together, to form a gigantic quantum wave. Once this wave has been created, nothing can affect it and there is no resistance at all. Even better, if you bring a magnet close to it, it will create a magnetic field which will make the superconducting wave whirl. This will then produce a magnetic field – rather like an electric coil – that repels the magnet, which will then actually levitate.

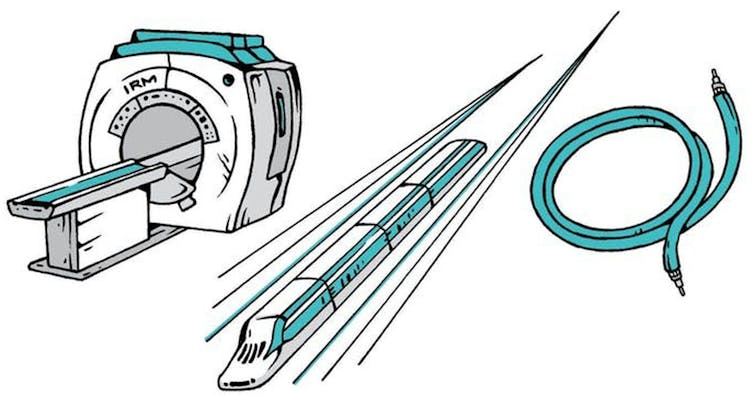
Some superconductors remain a mystery, even though more than a century has passed since their discovery. Cuprates, discovered in 1986, are currently the superconductors that work at the highest temperatures under ambient pressure. But we don’t know how electrons form the gigantic quantum wave mentioned above under those conditions. Understanding this remains one of the greatest challenges in today’s physics, at the heart of many research projects.
Could Kamerlingh Onnes ever have imagined that a simple electrical measurement in his noisy lab at Leiden would lead to one of the most rewarding areas of research?
This article was translated from the original French by Hélène Wilkinson.